
ENHANCING AND ENRICHING THERAPEUTIC OPTIONS IN PAEDIATRIC MEDICINE
Renaissance Pharma Limited, an Essential Pharma company, focused on development stage assets, is currently focused on improving treatment options within rare paediatric diseases. Our lead candidate – Daretabart (hu14.18K322A) is targeting the treatment of neuroblastoma.
Neuroblastoma
Neuroblastoma is a rare paediatric cancer arising from the abnormal development of cells in the sympathetic nervous system. It is the most common extracranial cancer in children and the leading cancer in children under one year of age, with an average age at diagnosis of 17 months1. Approximately half of all patients present with high-risk disease which has an overall survival rate of only 50% over five years, despite intensive therapy2,3. Neuroblastoma accounts for close to 10% of all childhood cancers and is the cause of 15% of all childhood cancer deaths2. There remains a clear need for safer and more effective treatment options, particularly for children with high-risk disease.
Daretabart (hu14.18K322A): an anti-GD2 therapy
IMPORTANT NOTICE: The information below relates to investigational compounds that are not approved for use in the UK, EU, or other markets unless specifically stated. This information is provided for corporate and scientific transparency only and is not intended to promote the use of any investigational product
Neuroblastomas abundantly express GD2, a molecule which inhibits recognition of cancer cells by immune cells, allowing the cancer to evade destruction by the body's immune system 5.
Renaissance Pharma, an Essential Pharma company, is developing Daretabart (hu14.18K322A) for the treatment of high-risk neuroblastoma. This investigational compound is currently being evaluated in clinical studies.
Daretabart (hu14.18K322A) is a humanised, anti-GD2 antibody immunotherapy engineered to target and destroy high-risk neuroblastoma (HRNB) cells. Its specifications include:
- 98% human composition
- Specialized cell line production with lower glycosylation of the antibody
- Engineered with a K322A mutation, limiting complement activation
Phase I and II trials have shown the treatment to be tolerable and clinically active in a variety of settings within HRNB.
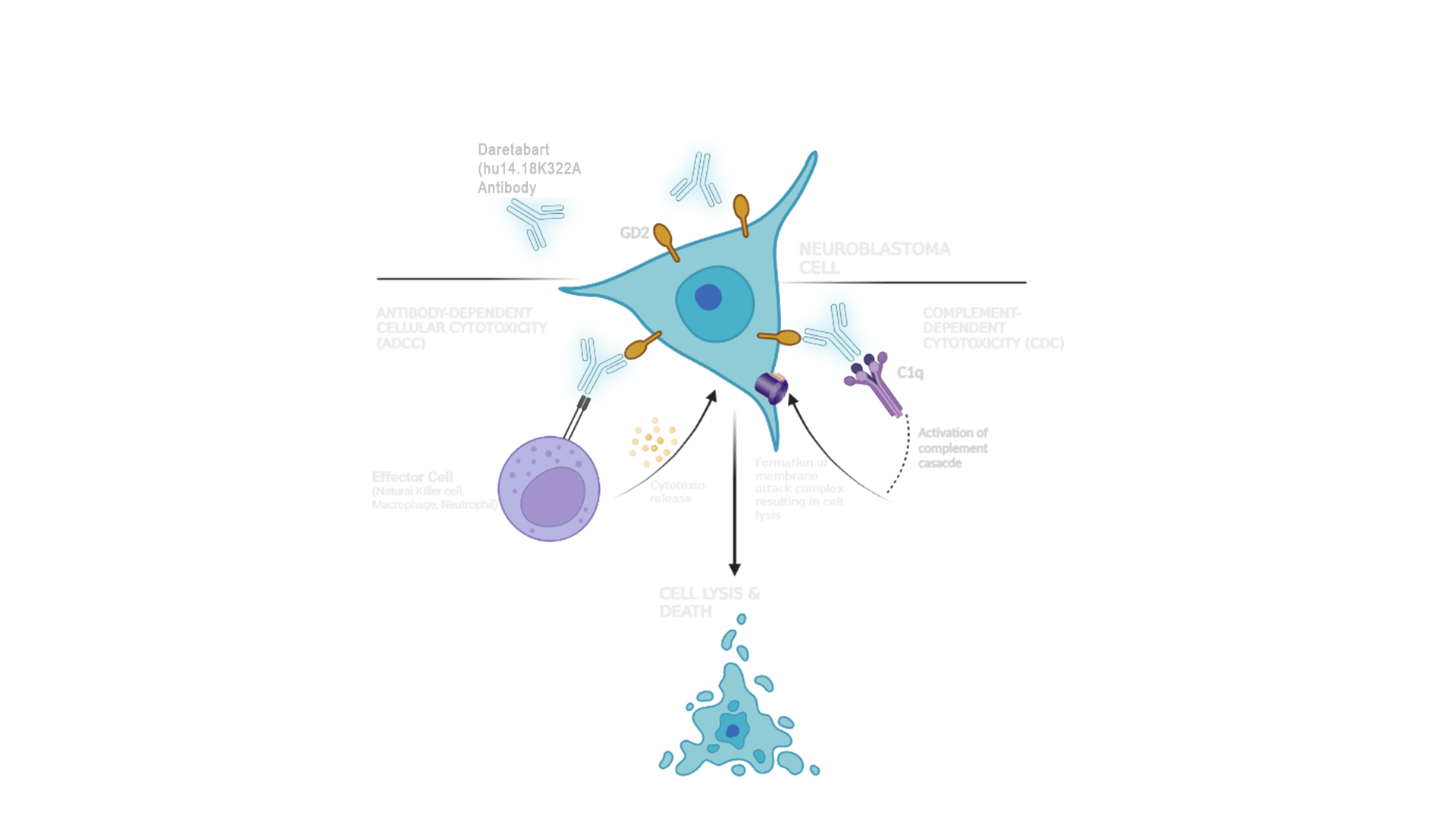
Image created with BioRender.com
References
Ries, L.A.G. ed., 1999. Cancer incidence and survival among children and adolescents: United States SEER program, 1975-1995 (No. 99). National Cancer Institute.
Whittle, S.B., Smith, V., Doherty, E., Zhao, S., McCarty, S. and Zage, P.E., 2017. Overview and recent advances in the treatment of neuroblastoma. Expert review of anticancer therapy, 17(4), pp.369-386.
Qiu, B. and Matthay, K.K., 2022. Advancing therapy for neuroblastoma. Nature Reviews Clinical Oncology, 19(8), pp.515-533.
Furman, W.L., 2021. Monoclonal antibody therapies for high risk neuroblastoma. Biologics: Targets and Therapy, pp.205-219.
Majzner R, 2022, Anti GD2 Antibody Therapy - an important approach in the treatment of neuroblastoma, Neuroblastoma Parent Global Symposium 2022 [Online]. Available from: https://www.youtube.com/watch?v=cSeN8-X5vQo [Accessed 25 February 2023]
Latest News
-

Renaissance Pharma Limited, an Essential Pharma company, secures FDA Fast Track Designation and IND clearance for Daretabart (hu1418K322A) in high-risk neuroblastoma
UK – 14 April 2026 – Renaissance Pharma Limited, an Essential Pharma company focused on development stage assets, today announces that the US Food and Drug Administration (FDA) has granted Fast Track Designation for Daretabart (hu1418K322A)…
-

Essential Pharma acquires Renaissance Pharma Ltd and its clinical stage immunotherapy for the treatment of high-risk neuroblastoma
April 9, 2024, UK | Essential Pharma, an international specialty pharma group focused on ensuring that patients have sustainable access to low volume, clinically differentiated, niche pharmaceutical products across key therapeutic areas, announces that it has completed the acquisition of the entire issued share capital of….
-

Renaissance Pharma launches and announces the in-licensing of Hu14.18K322A (Hu14.18) from St.Jude Children's Hospital for the treatment of High-Risk Neuroblastoma
August 1, 2023, Manchester, UK | Renaissance Pharma Ltd., a rapidly emerging company focused on the development of life changing therapies in paediatric rare disease, today launches and announces the signing of an exclusive license agreement….
